fractional rf microneedle machine exporter
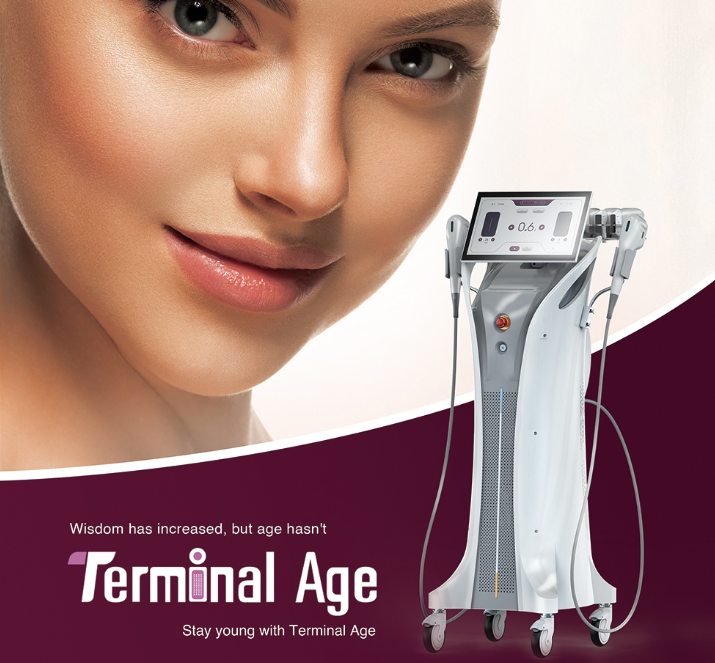
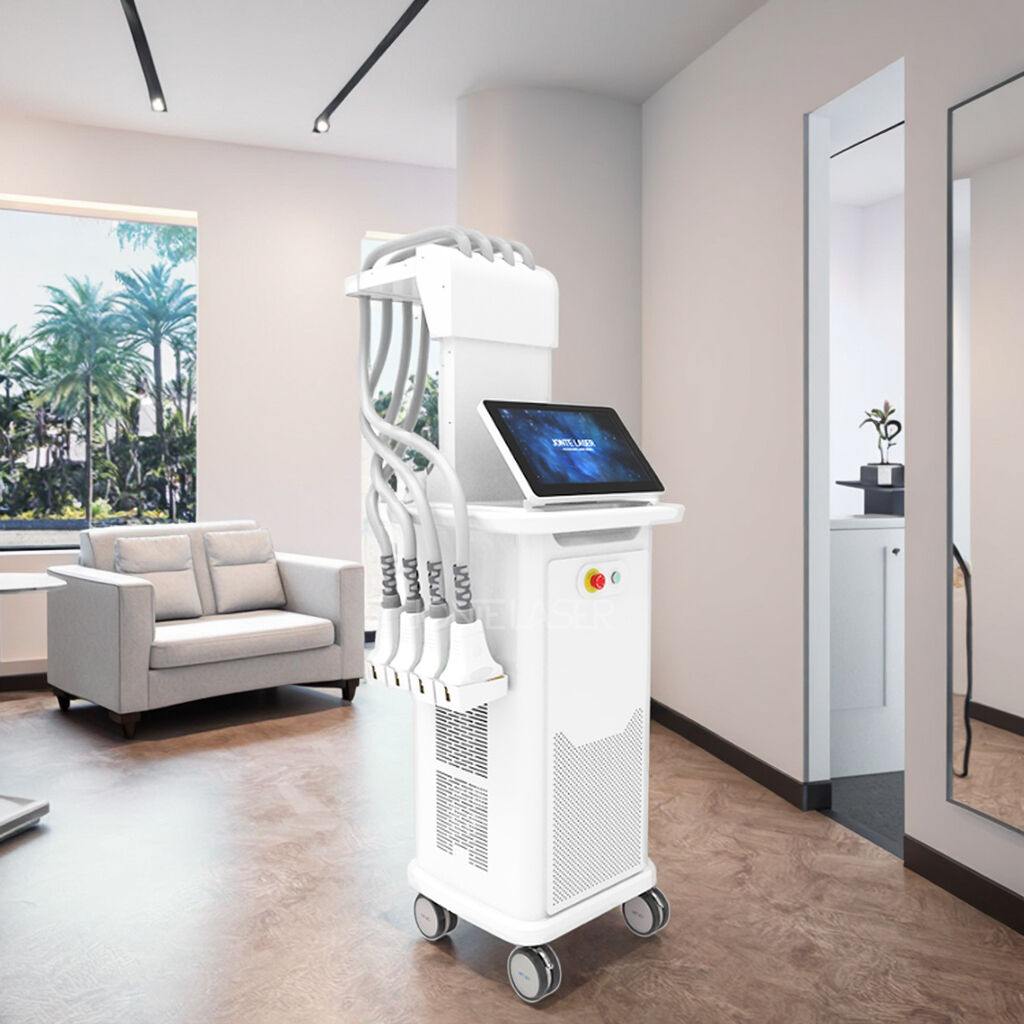
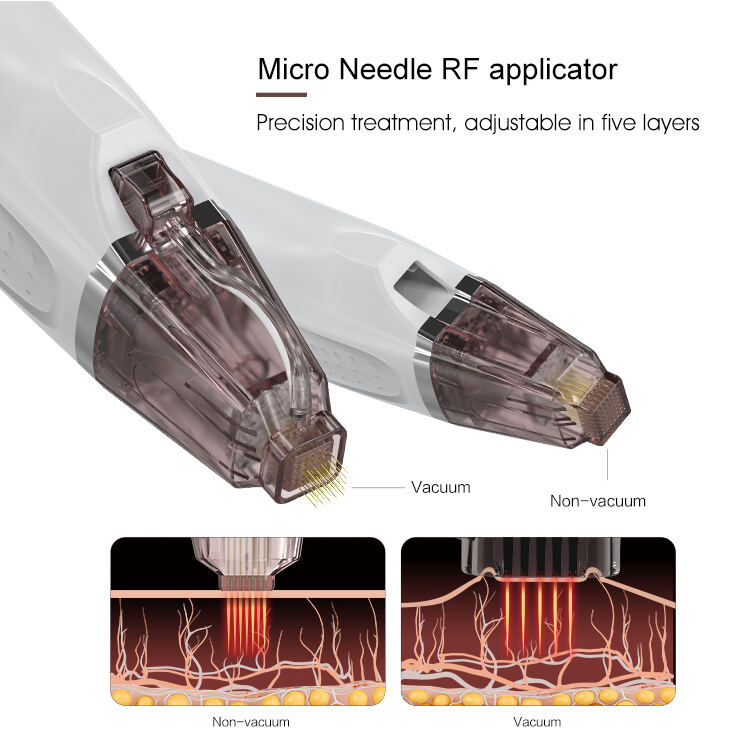
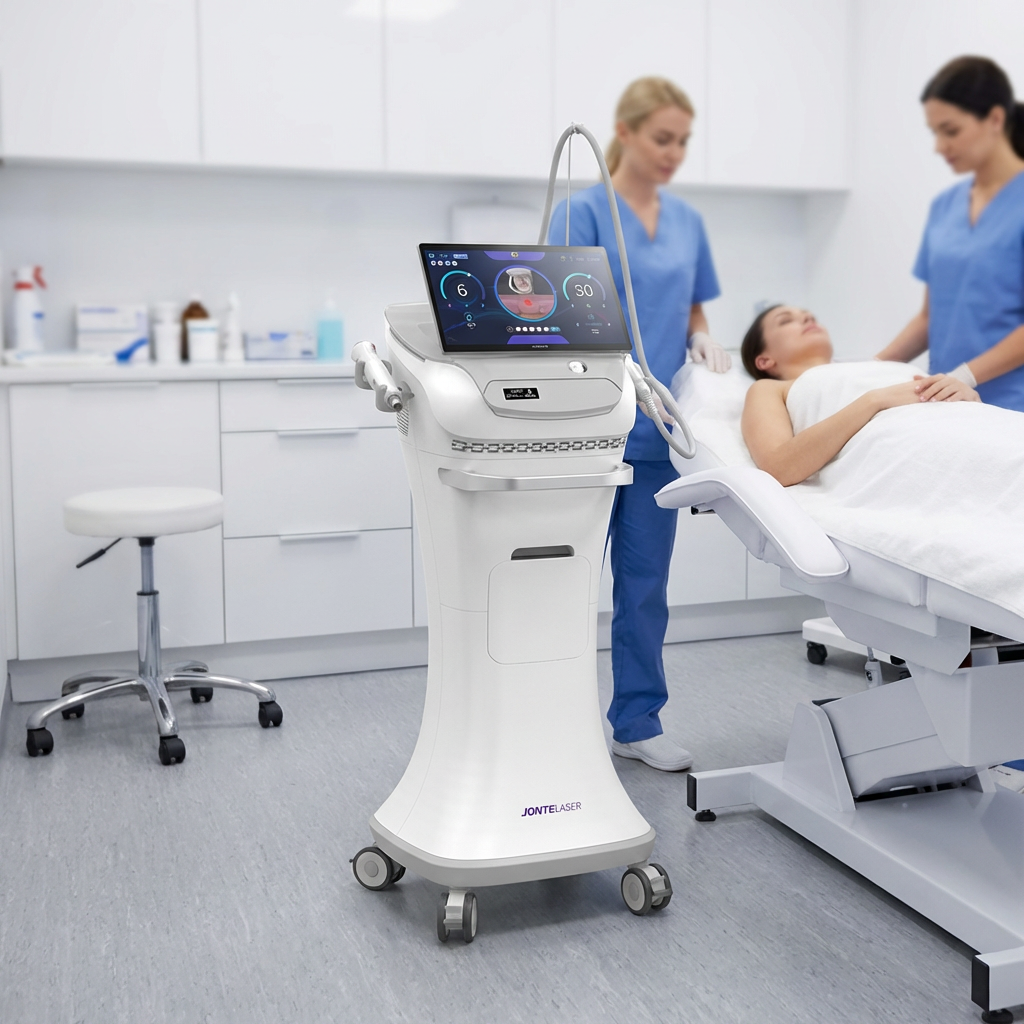
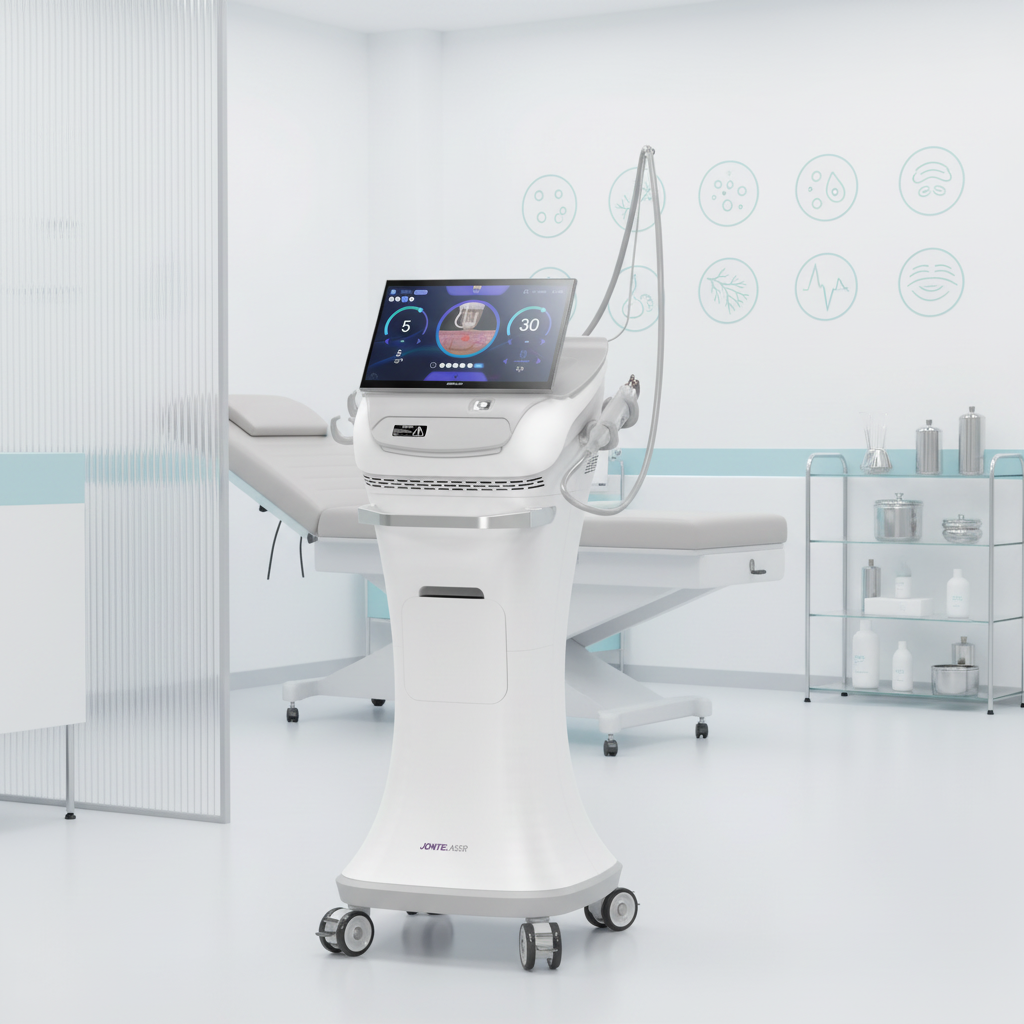
The fractional rf microneedle machine exporter represents a cutting-edge advancement in aesthetic technology, combining radiofrequency energy with precision microneedling capabilities. This sophisticated equipment delivers controlled thermal energy through ultra-fine needles that penetrate the skin at predetermined depths, creating microscopic channels while simultaneously heating dermal tissues. The fractional rf microneedle machine exporter incorporates advanced temperature monitoring systems and adjustable penetration depths, ranging from 0.5mm to 3.5mm, ensuring optimal treatment customization for various skin conditions. The device features multiple needle configurations, typically offering 25, 49, or 81-pin arrangements, allowing practitioners to select the most appropriate treatment pattern for specific areas and patient needs. Modern fractional rf microneedle machine exporter units integrate digital displays with intuitive interfaces, providing real-time feedback on treatment parameters including energy output, temperature readings, and penetration depth. The technology operates on the principle of fractional wound healing, where controlled micro-injuries stimulate natural collagen production and cellular regeneration. These machines incorporate safety mechanisms such as automatic shut-off systems, impedance monitoring, and temperature regulation to ensure consistent and safe treatment delivery. The fractional rf microneedle machine exporter typically includes disposable needle cartridges, ensuring sterility and preventing cross-contamination between patients. Advanced models feature cooling systems to enhance patient comfort during procedures and minimize thermal damage to surrounding tissues. The equipment supports various treatment protocols, from superficial skin rejuvenation to deeper structural remodeling, making it versatile for addressing multiple aesthetic concerns including acne scars, stretch marks, wrinkles, and skin laxity. Export models often comply with international medical device standards, including CE marking and FDA approvals, ensuring global market accessibility for distributors and clinics worldwide.