fractional rf microneedle machine factory
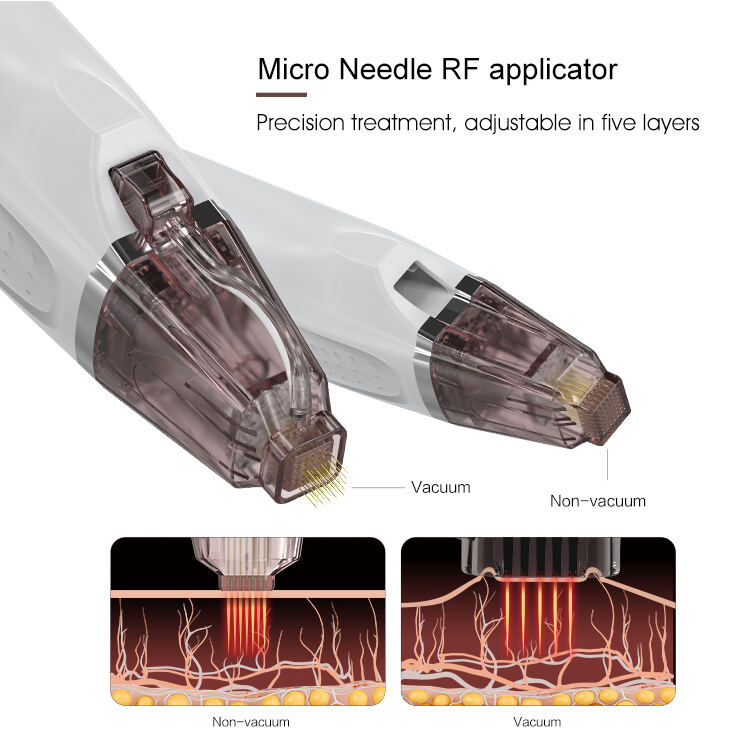
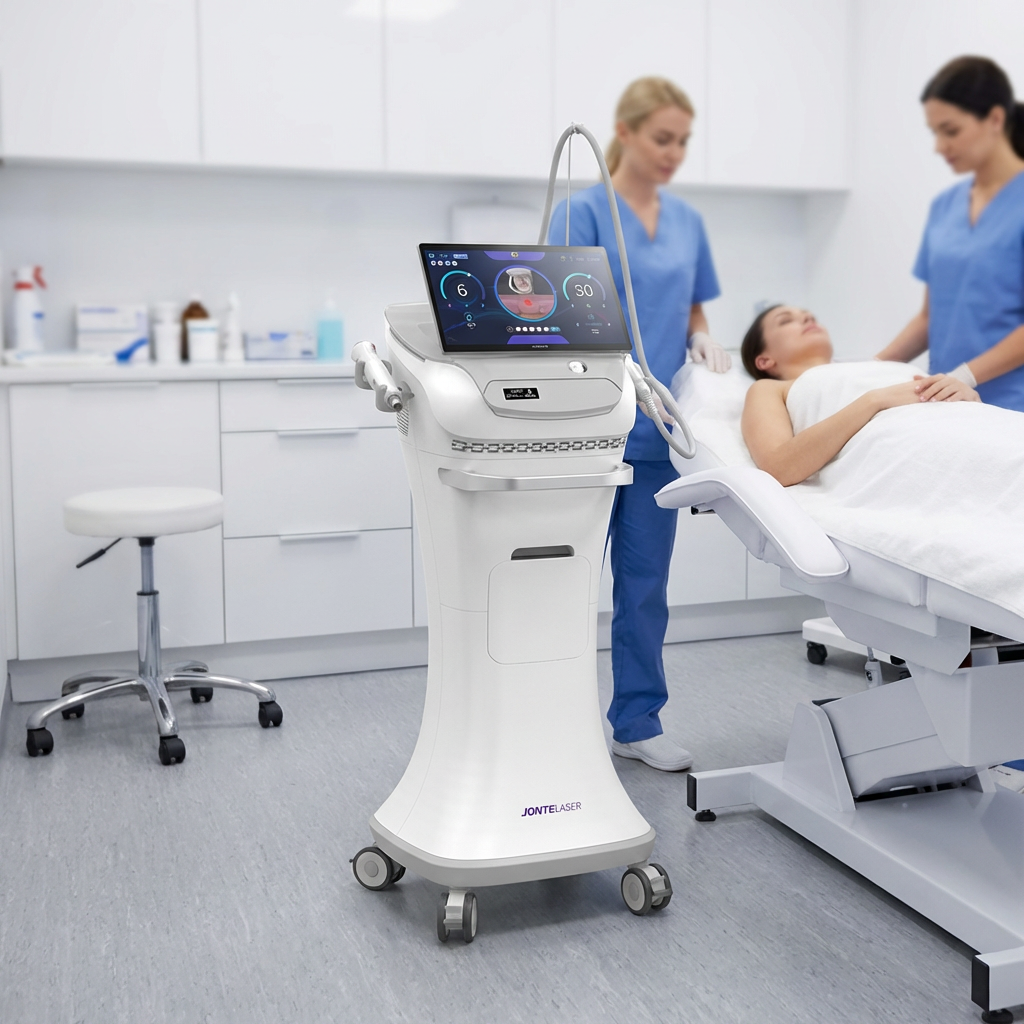
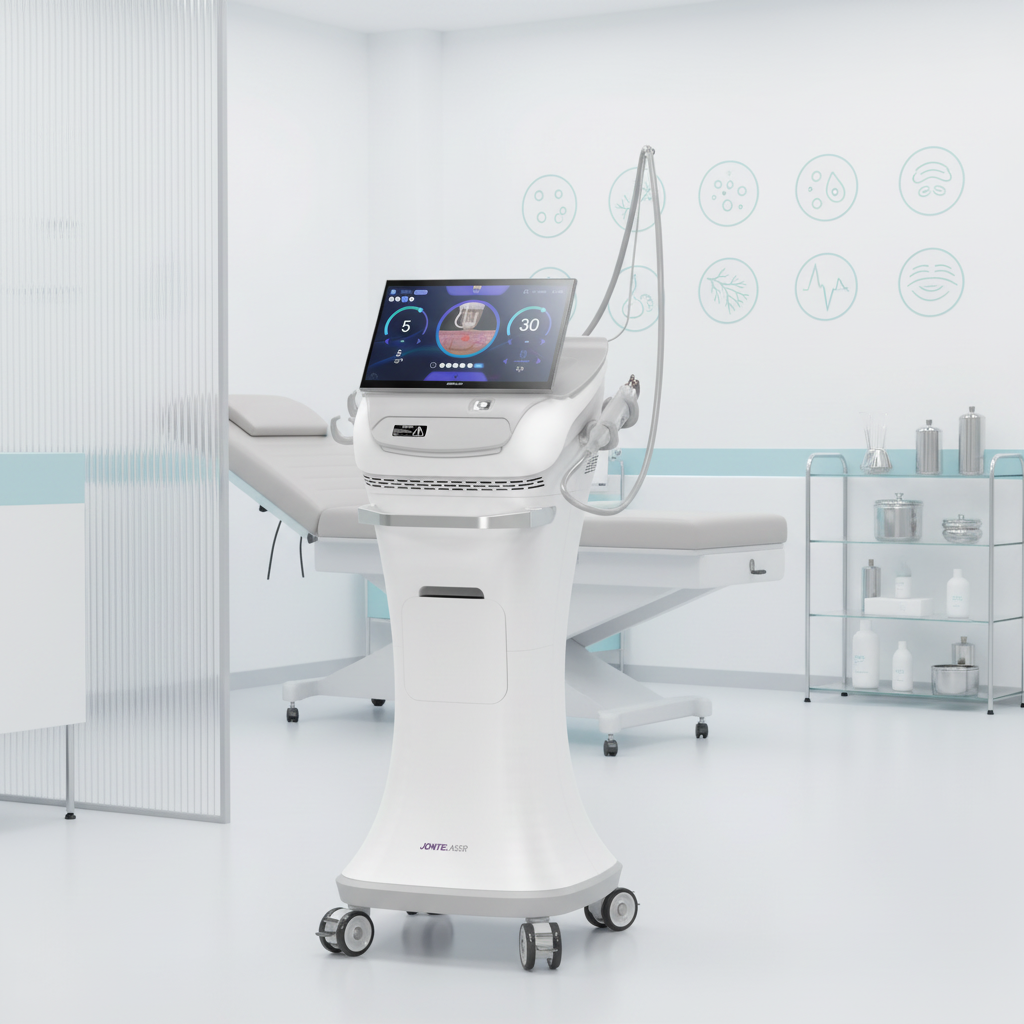
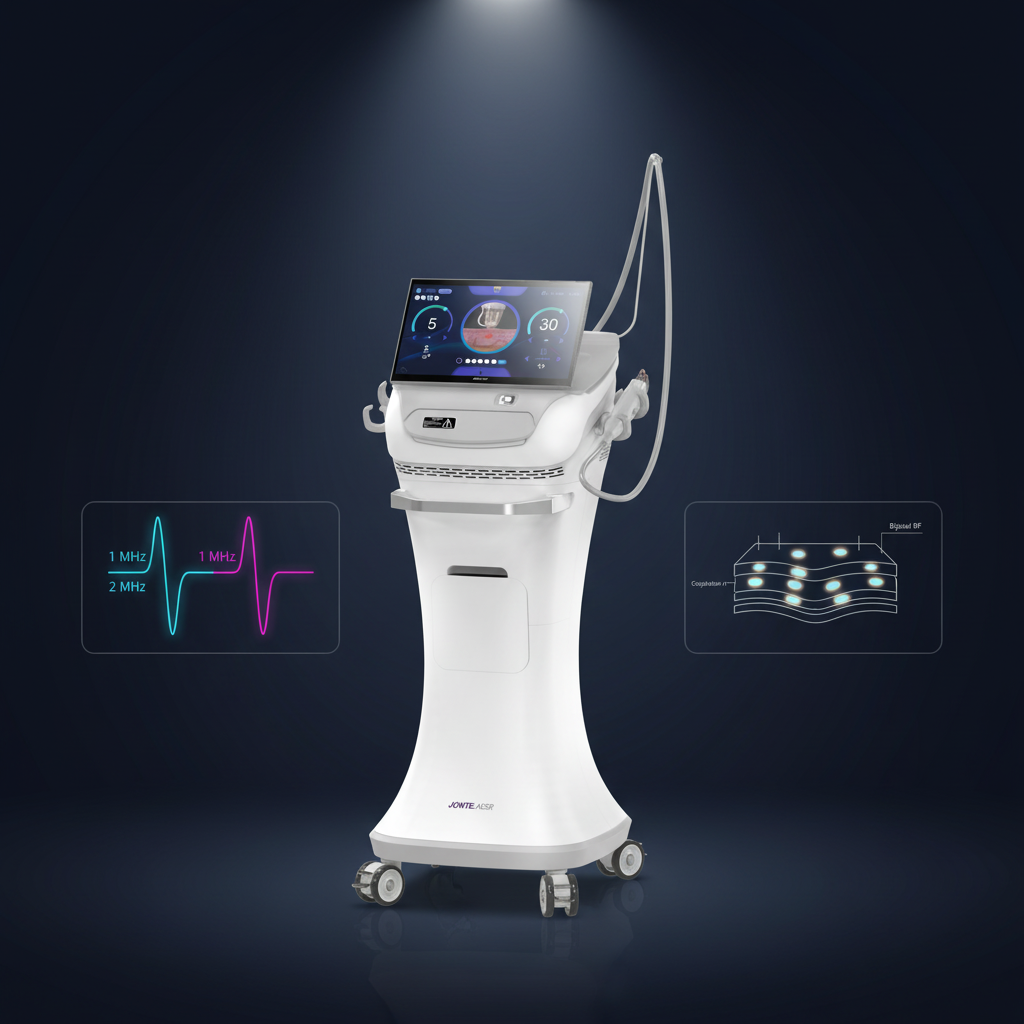
A fractional rf microneedle machine factory represents the pinnacle of modern aesthetic medical equipment manufacturing, combining cutting-edge radiofrequency technology with precision microneedling systems. These specialized manufacturing facilities produce advanced beauty devices that deliver controlled radiofrequency energy through ultra-fine needles, creating microscopic channels in the skin while simultaneously heating deeper tissue layers. The primary functions of equipment from a fractional rf microneedle machine factory include skin tightening, wrinkle reduction, scar treatment, and overall facial rejuvenation. The technological framework incorporates sophisticated control systems that regulate needle depth, energy output, and treatment patterns to ensure optimal results across diverse skin types and conditions. Manufacturing processes within a fractional rf microneedle machine factory emphasize precision engineering, quality assurance protocols, and adherence to international medical device standards. The machines produced feature adjustable needle configurations ranging from 0.5mm to 3.5mm depths, allowing practitioners to customize treatments for various skin concerns and anatomical areas. Advanced cooling systems integrated during manufacturing ensure patient comfort during procedures while maintaining consistent energy delivery. Applications extend beyond facial treatments to include body contouring, stretch mark reduction, and hair restoration procedures. The fractional rf microneedle machine factory environment maintains sterile production conditions, utilizing cleanroom technologies and rigorous testing protocols. Quality control measures include electromagnetic compatibility testing, biocompatibility assessments, and clinical validation studies. Modern factories incorporate automated assembly lines, precision calibration equipment, and comprehensive documentation systems to ensure each device meets exact specifications. The integration of digital interfaces allows practitioners to store treatment protocols, track patient progress, and maintain detailed treatment records. These manufacturing facilities continuously innovate, developing enhanced needle technologies, improved energy delivery systems, and user-friendly interfaces that streamline treatment procedures while maximizing patient safety and treatment efficacy.