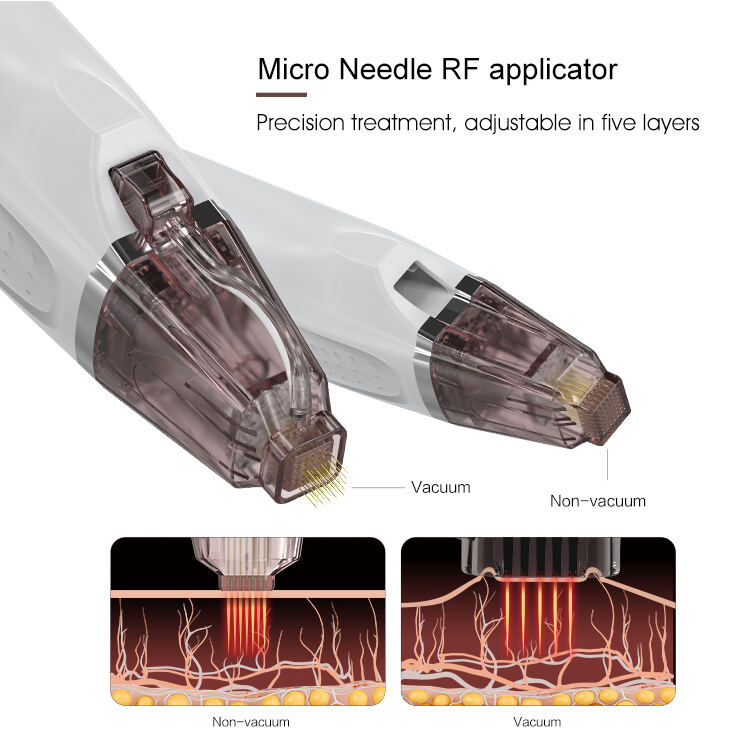
Regulatory Compliance Assurance and International Certification Management
Regulatory compliance assurance represents a critical service dimension that distinguishes professional fractional co2 laser machine exporters from simple equipment distributors, providing essential value for healthcare providers navigating complex international medical device regulations. The importance of proper regulatory compliance cannot be overstated in the medical device industry, where non-compliant equipment can result in significant legal liabilities, practice shutdowns, and professional license revocations that could permanently damage healthcare careers and business operations. A competent fractional co2 laser machine exporter maintains current knowledge of regulatory requirements across multiple jurisdictions, including FDA standards in the United States, CE marking requirements in Europe, Health Canada approvals, and various other national certification programs that govern medical device importation and clinical use. This expertise proves invaluable for healthcare providers who lack the resources or specialized knowledge necessary to navigate complex regulatory landscapes independently, particularly when expanding practices into new geographic markets or upgrading existing equipment to newer technology platforms. The certification management services provided by experienced exporters include documentation preparation, regulatory filing assistance, and ongoing compliance monitoring that ensures continued adherence to evolving safety standards and performance requirements throughout the equipment operational lifecycle. Professional fractional co2 laser machine exporters also maintain relationships with recognized testing laboratories and certification bodies, enabling expedited approval processes and reducing the time required to bring new equipment into clinical service. Quality assurance programs implemented by reputable exporters include rigorous pre-shipment testing protocols, comprehensive documentation packages, and traceability systems that facilitate rapid response to any safety concerns or regulatory inquiries that may arise after equipment deployment. The value of regulatory compliance assurance extends beyond simple legal protection to encompass enhanced credibility with patients, insurance providers, and professional colleagues who recognize the importance of operating only certified and properly approved medical equipment. Risk mitigation strategies employed by professional exporters include comprehensive insurance coverage, warranty programs, and recall management procedures that protect healthcare providers from potential financial losses associated with equipment defects or regulatory changes that might affect device approval status.